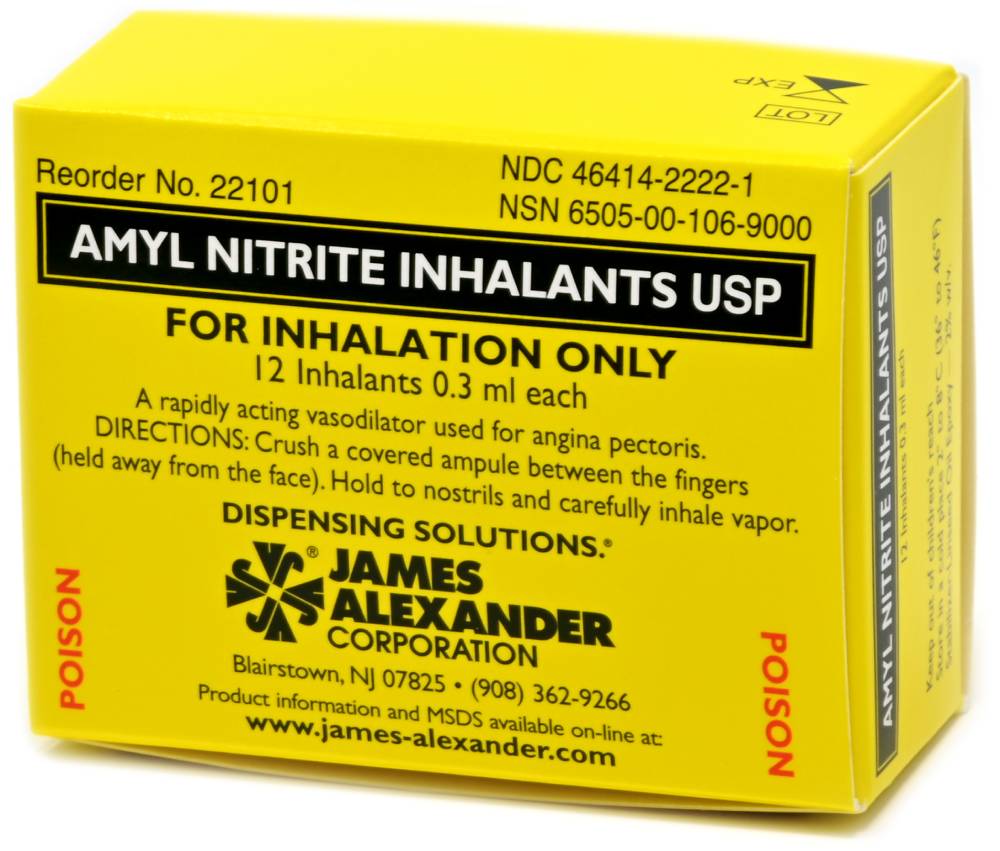
Indications
Amyl Nitrite Inhalant USP is an organic nitrite used for the relief and prophylactic management of angina pectoris. The drug is delivered via inhalation.
Pharmacology
The principal pharmacological action of Amyl Nitrite is relaxation of vascular smooth muscle, resulting in generalized vasodilation. Although Amyl Nitrite reflexly increases heart rate and myocardial contractility which increases myocardial oxygen consumption, the reduction in ventricular wall tension results in a net decrease in myocardial oxygen consumption. By decreasing myocardial oxygen consumption, Amyl Nitrite alters the imbalance of myocardial oxygen supply which causes angina pectoris.
Product Stability
James Alexander’s Amyl Nitrite Inhalant USP carries an expiration date of four years.
Product Storage Conditions
Keep protected from light at 2-8 degrees C (36-46 degrees F). Amyl Nitrite is extremely flammable. Keep from heat and open flame.
Directions for Use
The inhalant should be held away from the face while activating the package. To break the inner vial, crush ONCE between the thumb and forefinger at the dot located at the center of the inhalant. Carefully pass the inhalant back and forth close to the nose of the affected person several times. Discard inhalant after use. Relief usually occurs in one to three minutes.
Precautions
Amyl Nitrite Inhalant is a prescription drug. Use only in accordance with a physician’s instructions. FDA regulations specifically prohibit labeling the product for uses other than the treatment of angina pectoris (see 21CFR 250.102)
James Alexander’s Product Configuration and Packaging
- James Alexander’s Code 22101 – 12 individual boxes of 12 inhalants packaged in a carton. Packaged 264 boxes per shipping
For smaller order quantities, please contact Caligor Rx
Caligor Rx Inc. – (212)-988-1729 – inquiries@caligorpharmacy.com


 DIRECTIONS
DIRECTIONS